Indications & Important Safety Information
Indications
Important Safety Information
Indications
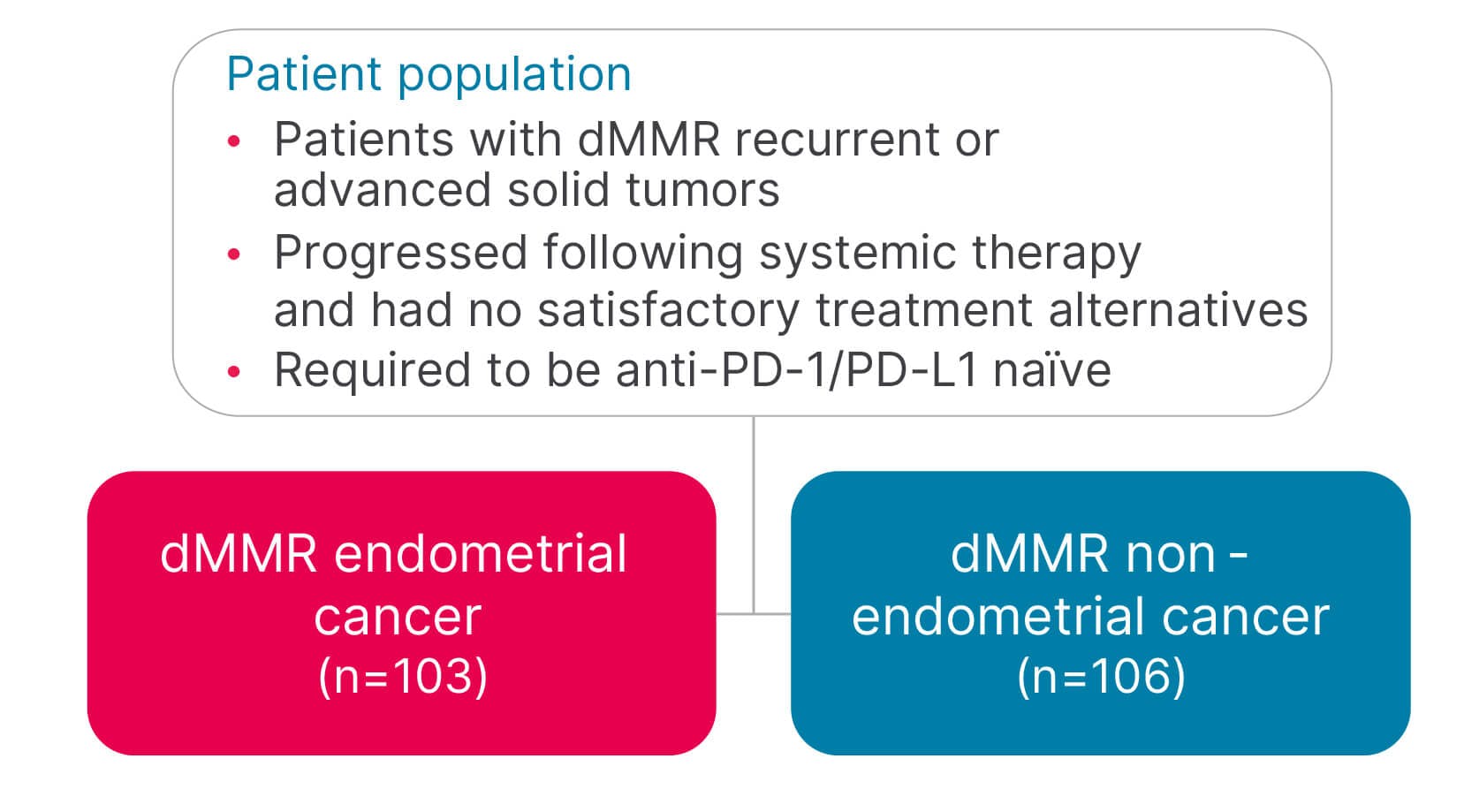
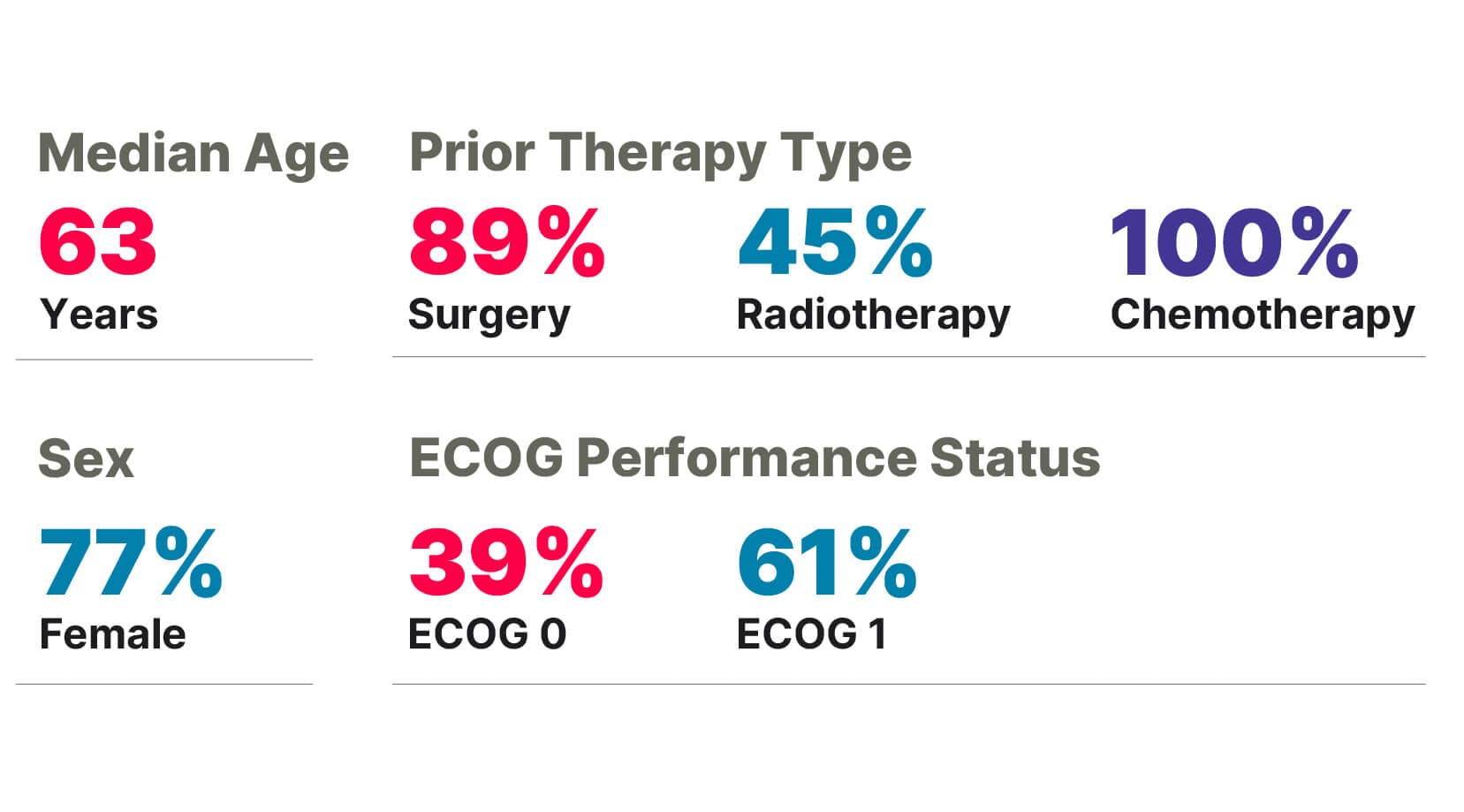
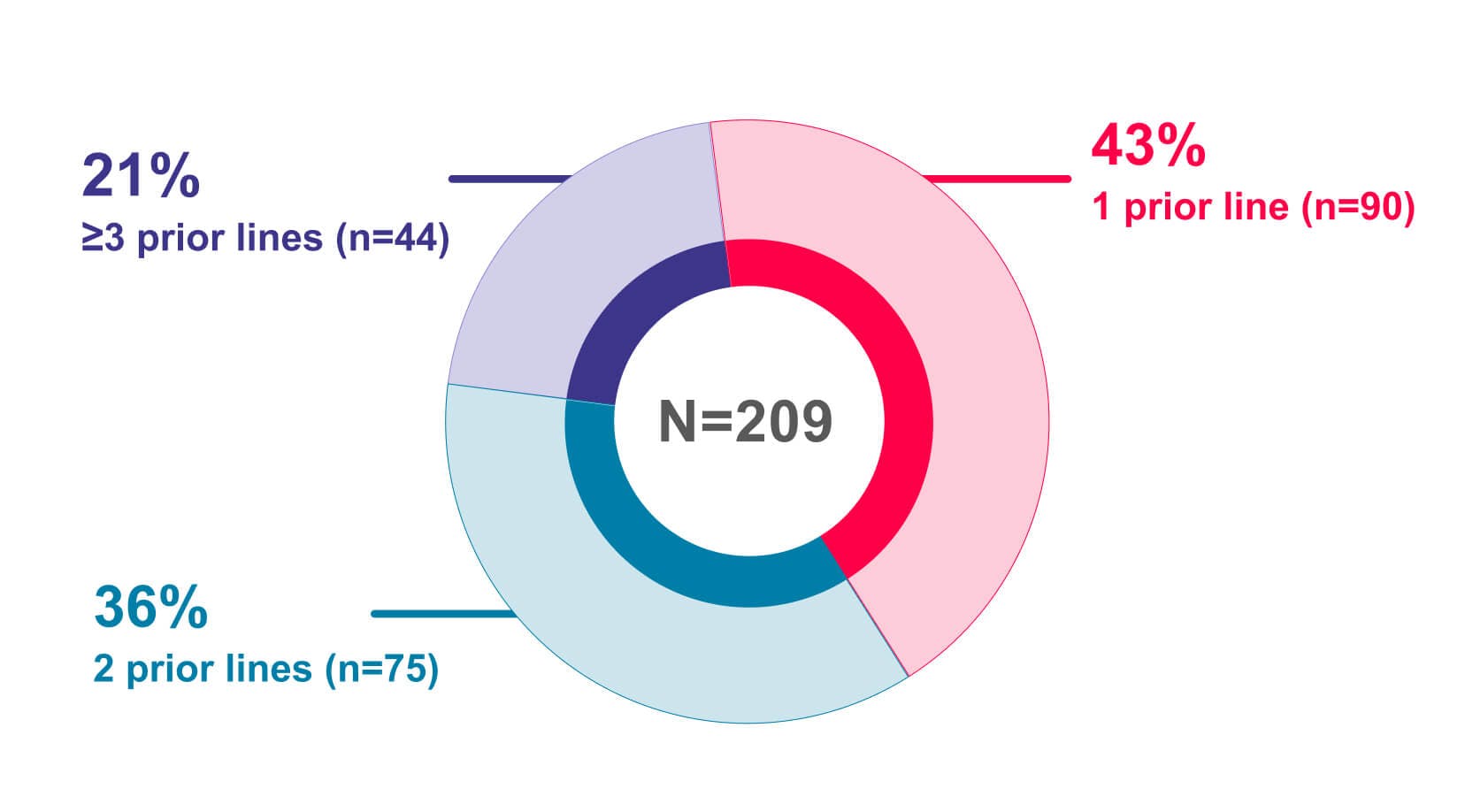
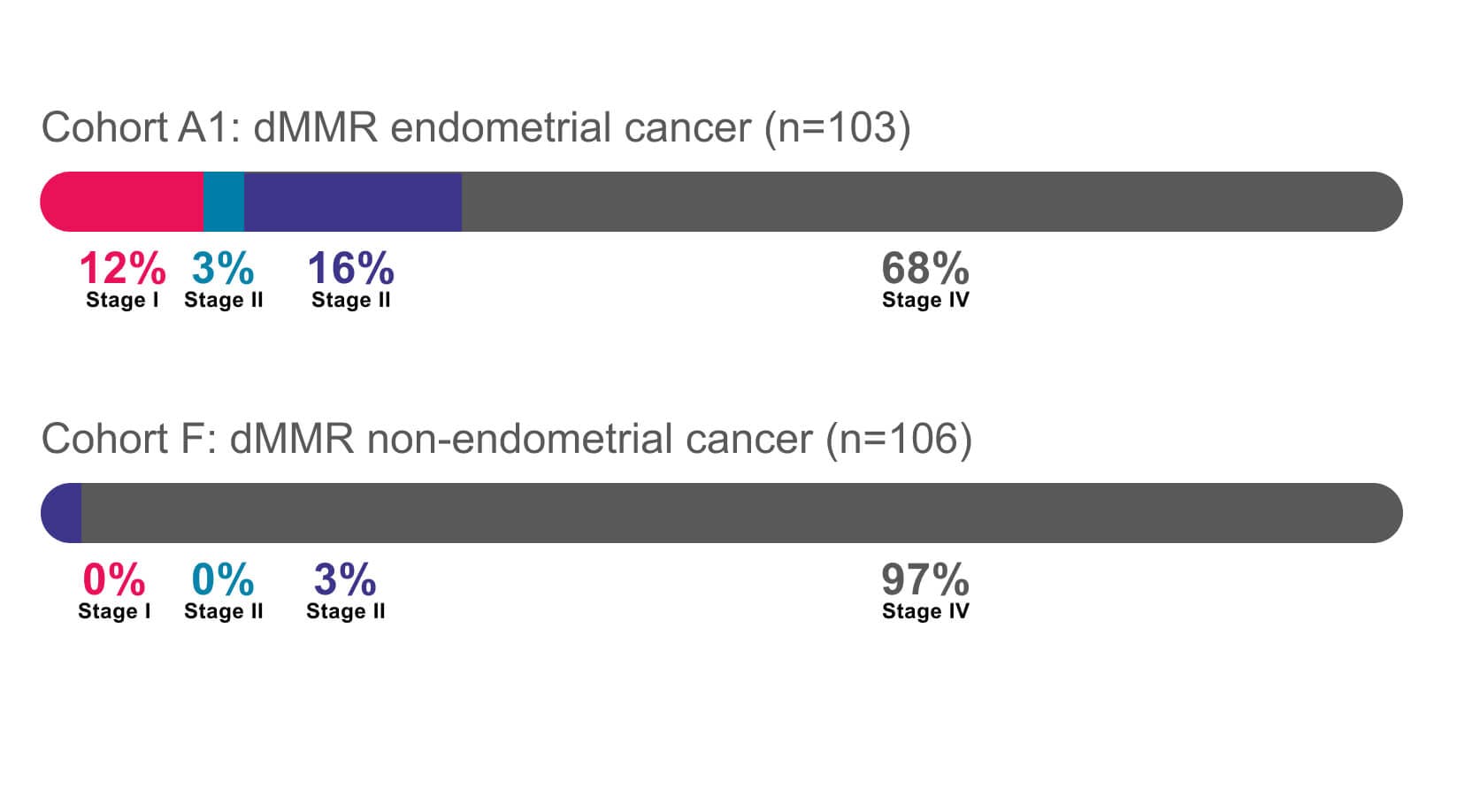
- JEMPERLI, in combination with carboplatin and paclitaxel, followed by JEMPERLI as a single agent, is indicated for the treatment of adult patients with primary advanced or recurrent endometrial cancer (EC).
- JEMPERLI, as a single agent, is indicated for the treatment of adult patients with mismatch repair deficient (dMMR) recurrent or advanced:
- EC, as determined by an FDA-approved test, that has progressed on or following prior treatment with a platinum-containing regimen in any setting and are not candidates for curative surgery or radiation, or
- solid tumors, as determined by an FDA-approved test, that have progressed on or following prior treatment and who have no satisfactory alternative treatment options. This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
Important Safety Information
- Immune-mediated adverse reactions, which can be severe or fatal, can occur in any organ system or tissue and can occur at any time during or after treatment with a PD-1/PD-L1–blocking antibody, including JEMPERLI.
- Monitor closely for signs and symptoms of immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function tests at baseline and periodically during treatment. For suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
- Based on the severity of the adverse reaction, withhold or permanently discontinue JEMPERLI. In general, if JEMPERLI requires interruption or discontinuation, administer systemic corticosteroids (1 to 2 mg/kg/day prednisone or equivalent) until improvement to ≤Grade 1. Upon improvement to ≤Grade 1, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reaction is not controlled with corticosteroids.
Immune-Mediated Pneumonitis
- JEMPERLI can cause immune-mediated pneumonitis, which can be fatal. In patients treated with other PD-1/PD-L1–blocking antibodies, the incidence of pneumonitis is higher in patients who have received prior thoracic radiation. Pneumonitis occurred in 2.3% (14/605) of patients, including Grade 2 (1.3%), Grade 3 (0.8%), and Grade 4 (0.2%) pneumonitis.
Immune-Mediated Colitis
- Colitis occurred in 1.3% (8/605) of patients, including Grade 2 (0.7%) and Grade 3 (0.7%) adverse reactions. Cytomegalovirus infection/reactivation have occurred in patients with corticosteroid-refractory immune-mediated colitis. In such cases, consider repeating infectious workup to exclude alternative etiologies.
Immune-Mediated Hepatitis
- JEMPERLI can cause immune-mediated hepatitis, which can be fatal. Grade 3 hepatitis occurred in 0.5% (3/605) of patients.
Immune-Mediated Endocrinopathies
- Adrenal Insufficiency
- Adrenal insufficiency occurred in 1.2% (7/605) of patients, including Grade 2 (0.5%) and Grade 3 (0.7%). For Grade 2 or higher adrenal insufficiency, initiate symptomatic treatment per institutional guidelines, including hormone replacement as clinically indicated. Withhold or permanently discontinue JEMPERLI depending on severity.
- Hypophysitis
- JEMPERLI can cause immune-mediated hypophysitis. Grade 3 hypophysitis occurred in 0.4% (1/241) of patients receiving JEMPERLI in combination with carboplatin and paclitaxel. Grade 2 hypophysitis occurred in 0.2% (1/605) of patients receiving JEMPERLI as a single agent. Initiate hormone replacement as clinically indicated. Withhold or permanently discontinue JEMPERLI depending on severity.
- JEMPERLI can cause immune-mediated hypophysitis. Grade 3 hypophysitis occurred in 0.4% (1/241) of patients receiving JEMPERLI in combination with carboplatin and paclitaxel. Grade 2 hypophysitis occurred in 0.2% (1/605) of patients receiving JEMPERLI as a single agent. Initiate hormone replacement as clinically indicated. Withhold or permanently discontinue JEMPERLI depending on severity.
- Thyroid Disorders
- Grade 2 thyroiditis occurred in 0.5% (3/605) of patients. Grade 2 hypothyroidism occurred in 12% (30/241) of patients receiving JEMPERLI in combination with carboplatin and paclitaxel. Grade 2 hypothyroidism occurred in 8% (46/605) of patients receiving JEMPERLI as a single agent. Hyperthyroidism occurred in 3.3% (8/241) of patients receiving JEMPERLI in combination with carboplatin and paclitaxel, including Grade 2 (2.9%) and Grade 3 (0.4%). Hyperthyroidism occurred in 2.3% (14/605) of patients receiving JEMPERLI as a single agent, including Grade 2 (2.1%) and Grade 3 (0.2%). Initiate thyroid hormone replacement or medical management of hyperthyroidism as clinically indicated. Withhold or permanently discontinue JEMPERLI depending on severity.
- Type 1 Diabetes Mellitus, Which Can Present with Diabetic Ketoacidosis
- JEMPERLI can cause type 1 diabetes mellitus, which can present with diabetic ketoacidosis. Grade 3 type 1 diabetes mellitus occurred in 0.4% (1/241) of patients receiving JEMPERLI in combination with carboplatin and paclitaxel. Grade 3 type 1 diabetes mellitus occurred in 0.2% (1/605) of patients receiving JEMPERLI as a single agent. Monitor patients for hyperglycemia or other signs and symptoms of diabetes. Initiate treatment with insulin as clinically indicated. Withhold or permanently discontinue JEMPERLI depending on severity.
Immune-Mediated Nephritis with Renal Dysfunction
- JEMPERLI can cause immune-mediated nephritis, which can be fatal. Grade 2 nephritis, including tubulointerstitial nephritis, occurred in 0.5% (3/605) of patients.
Immune-Mediated Dermatologic Adverse Reactions
- JEMPERLI can cause immune-mediated rash or dermatitis. Bullous and exfoliative dermatitis, including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug rash with eosinophilia and systemic symptoms (DRESS), have occurred with PD-1/PD-L1–blocking antibodies. Topical emollients and/or topical corticosteroids may be adequate to treat mild to moderate non-bullous/exfoliative rashes. Withhold or permanently discontinue JEMPERLI depending on severity.
Other Immune-Mediated Adverse Reactions
- The following clinically significant immune-mediated adverse reactions occurred in <1% of the 605 patients treated with JEMPERLI or were reported with the use of other PD-1/PD-L1–blocking antibodies. Severe or fatal cases have been reported for some of these adverse reactions.
- Nervous System: Meningitis, encephalitis, myelitis and demyelination, myasthenic syndrome/myasthenia gravis, Guillain-Barré syndrome, nerve paresis, autoimmune neuropathy
- Cardiac/Vascular: Myocarditis, pericarditis, vasculitis
- Ocular: Uveitis, iritis, other ocular inflammatory toxicities. Some cases can be associated with retinal detachment. Various grades of visual impairment to include blindness can occur
- Gastrointestinal: Pancreatitis, including increases in serum amylase and lipase levels, gastritis, duodenitis
- Musculoskeletal and Connective Tissue: Myositis/polymyositis, rhabdomyolysis and associated sequelae including renal failure, arthritis, polymyalgia rheumatica
- Endocrine: Hypoparathyroidism
- Other (Hematologic/Immune): Autoimmune hemolytic anemia, aplastic anemia, hemophagocytic lymphohistiocytosis, systemic inflammatory response syndrome, histiocytic necrotizing lymphadenitis (Kikuchi lymphadenitis), sarcoidosis, immune thrombocytopenia, solid organ transplant rejection, other transplant (including corneal graft) rejection
Infusion-Related Reactions
- Severe or life-threatening infusion-related reactions have been reported with PD-1/PD-L1–blocking antibodies. Severe infusion-related reactions (Grade 3) occurred in 0.2% (1/605) of patients receiving JEMPERLI. Monitor patients for signs and symptoms of infusion-related reactions. Interrupt or slow the rate of infusion or permanently discontinue JEMPERLI based on severity of reaction.
Complications of Allogeneic HSCT
- Fatal and other serious complications can occur in patients who receive allogeneic hematopoietic stem cell transplantation (HSCT) before or after treatment with a PD-1/PD-L1–blocking antibody, which may occur despite intervening therapy. Monitor patients closely for transplant-related complications and intervene promptly.
Embryo-Fetal Toxicity and Lactation
- Based on its mechanism of action, JEMPERLI can cause fetal harm. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with JEMPERLI and for 4 months after their last dose. Because of the potential for serious adverse reactions from JEMPERLI in a breastfed child, advise women not to breastfeed during treatment with JEMPERLI and for 4 months after their last dose.
Common Adverse Reactions
The most common adverse reactions (≥20%), including laboratory abnormalities, in patients with EC who received JEMPERLI in combination with carboplatin and paclitaxel were decreased hemoglobin, increased creatinine, peripheral neuropathy, decreased white blood cell count, fatigue, nausea, alopecia, decreased platelets, increased glucose, decreased lymphocytes, decreased magnesium, decreased neutrophils, increased AST, arthralgia, rash, constipation, diarrhea, increased ALT, decreased potassium, decreased albumin, decreased sodium, increased alkaline phosphatase, abdominal pain, dyspnea, decreased appetite, increased amylase, decreased phosphate, urinary tract infection, and vomiting.
The most common adverse reactions (≥20%) in patients with dMMR EC who received JEMPERLI as a single agent were fatigue/asthenia, anemia, nausea, diarrhea, constipation, vomiting, and rash. The most common Grade 3 or 4 laboratory abnormalities (>2%) were decreased lymphocytes, decreased sodium, increased alanine aminotransferase, increased creatinine, decreased neutrophils, decreased albumin, and increased alkaline phosphatase.
The most common adverse reactions (≥20%) in patients with dMMR solid tumors who received JEMPERLI as a single agent were fatigue/asthenia, anemia, diarrhea, and nausea. The most common Grade 3 or 4 laboratory abnormalities (≥2%) were decreased lymphocytes, decreased sodium, increased alkaline phosphatase, and decreased albumin.
Please see full Prescribing Information, including Medication Guide.